Micro C Heads to World’s Largest Meeting of Orthopaedic Surgeons with Funding Boost to $2.2 Million
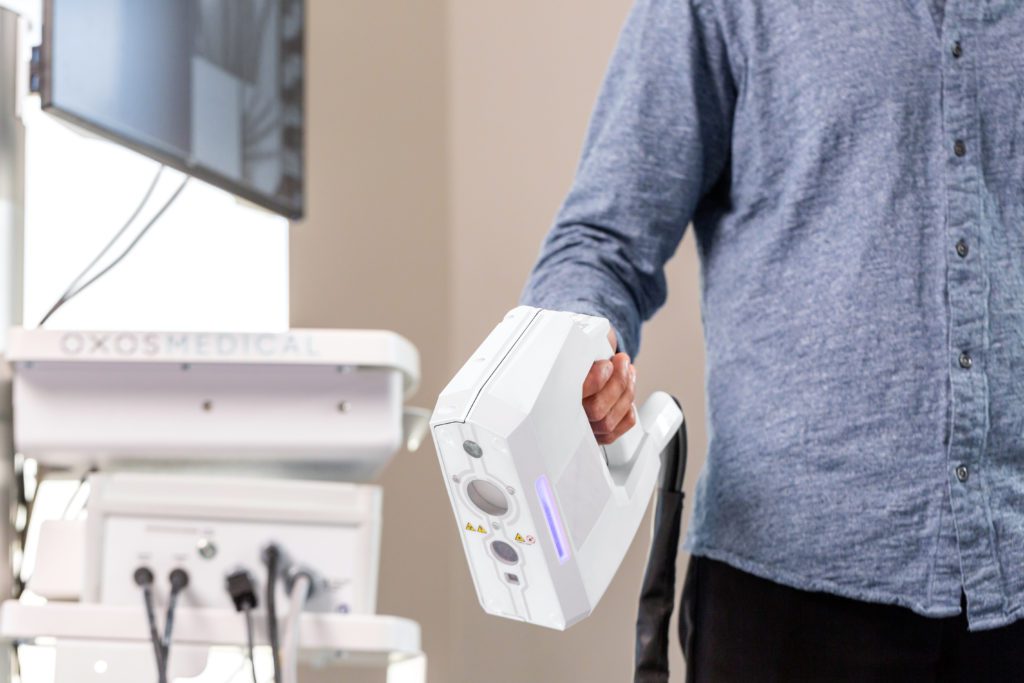
The Micro C team announces a $1 million add to its seed investment, now $2.2 million, on its way to the annual meeting of the 39,000-member American Academy of Orthopaedic Surgeons (AAOS). Micro C Imaging is poised to achieve its two major 2018 objectives: FDA compliance and commercial market launch of its revolutionary mobile fluoroscopy solution.
The company will educate members of the American Academy of Orthopaedic Surgeons, called Fellows, who are orthopaedists engaged in the diagnosis, care, and treatment of musculoskeletal disorders. 15,000 Fellows will attend the AAOS annual meeting taking place March 7-9 at the Morial Convention Center in New Orleans, LA . At its Booth #7018 in the First Time Exhibitors Hall, the Micro C team will educate orthopaedists, its initial target market, about the uses and benefits of the Micro C™.
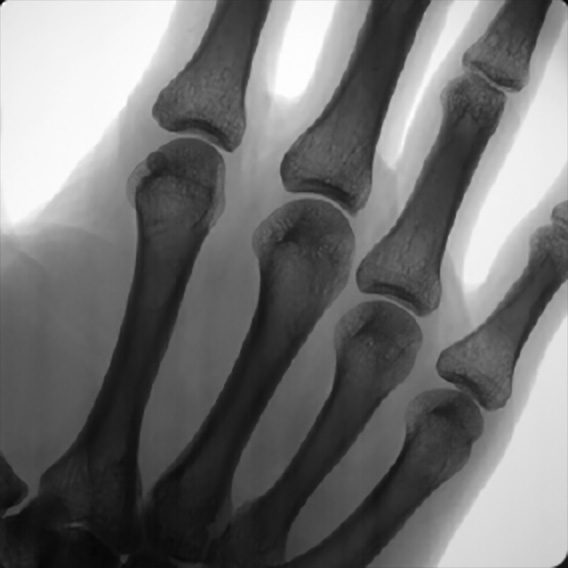
The Micro C is a handheld X-ray and digital camera that is ergonomically designed for surgeons and physicians treating extremities in both clinical and surgical settings. Micro C allows doctors to capture images with greater accuracy, clarity, safety, and speed. It incorporates a still, video and infrared camera, together with an image receptor and software enabling HIPPA-compliant image and data capture and electronic medical records (EMR) system integration. The clinical embodiment brings breakthrough mobile X-ray capability into physicians’ offices and clinics, urgent care facilities, emergency rooms, rural medicine, sports medicine, military field settings, and global health. The surgical embodiment enables surgeons treating disorders of the extremities to reduce operative time, thus allowing hospitals and surgery centers to greatly increase the utilization rate of their facilities and freeing up valuable surgical time to execute additional procedures.
“I greatly look forward to demonstrating the Micro C to my peers,” states Dr. Gregory Kolovich, Chief Medical Officer and Co-Founder. “I invented the Micro C to solve my own real world issues with the design of current orthopedic imaging devices that are bulky, slow, large and cumbersome. At AAOS we will share results from our testing programs that document the extraordinary decrease in radiation emission with the Micro C as compared with devices currently and widely in use.” The Micro C does not require any setup time, as the procedure is executed on the device directly and integrates into all compliant FHIR EMR systems, eliminating a major hurdle to HIPPA compliant image documentation.

Evan Ruff, CEO and Co-Founder, explains “Micro C represents a clean sheet design of the x-ray imaging device, combining a highly agile X-ray system with state-of-the-art visible and infrared imaging cameras.” He comments further that “We have captured our groundbreaking innovations in our pending patents which are in the USPTO’s Track One prioritized examination process, also known as the ‘fast track’.” Dr. Youssra Marjoua, Micro C’s International Medicine Chief, adds, “We especially will welcome the international orthopaedic surgeons, one-third of the physician attendees, to share the company’s keen interest in bringing the Micro C to international markets.”Uses of the Micro C include bringing X-ray capabilities for the first time into the highly challenging environments of rural and emergency healthcare.
Where both she and Dr. Kolovich have served as volunteer orthopaedic surgeons during missions and exchanges.